|
Three experiments led him to this.: irst, in a variation of an 1895 experiment by Jean Perrin, Thomson built a cathode ray tube ending in a pair of metal cylinders with a. 
These subatomic particles can be found within atoms of all elements.Ĭonclusion - The cathode rays are made up of negatively charged particles known as electrons. He advanced the idea that cathode rays are really streams of very small pieces of atoms. Thomson also placed two magnets on either side of the tube, and observed that this magnetic field also deflected the cathode ray. This indicated that the cathode ray was composed of negatively-charged particles. The Cathode ray experiment is based on conduction of electricity through gases at low pressure in a discharge tube. The cathode ray was deflected away from the negatively-charged electric plate and towards the positively-charged plate. 1) with slits in them are placed in a bulb connected with the discharge-tube the cathode rays from the cathode A pass into the bulb through a slit in a metal. Thomsons cathode ray experiment a sketch of JJ Thomson. Thomson used a cathode ray tube to determine the nature of the. Thomson's experiment on constituents of cathode rays:Įxperiment - To test the properties of the particles, Thomson placed two oppositely-charged electric plates around the cathode ray. Describe early milestones in the development of modern atomic theory. The Thompson Cathode Ray Experiment was an experiment conducted by J. An electric discharge is passed through the tube.Įmission of blue rays can be seen from the cathode.Ĭonclusion - The blue rays emitted from cathode are cathode rays. 
William Crookes experiment for the discovery of cathode rays:Įxperiment - The figure shows a vacuum tube which has a gas at a very low pressure. 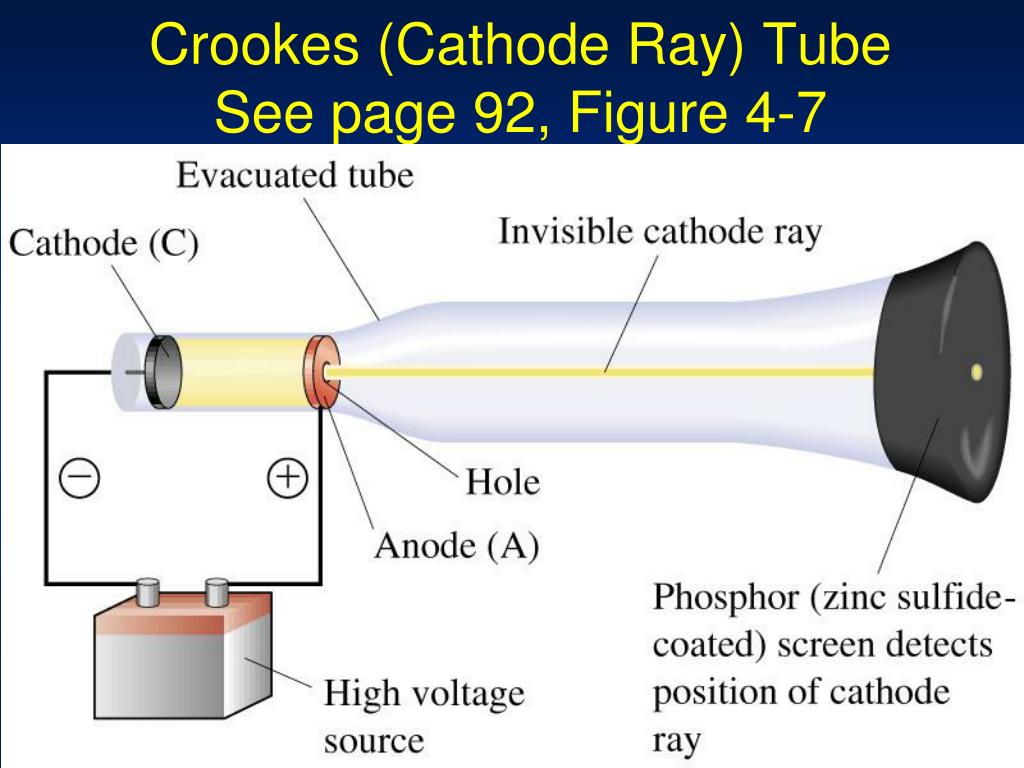
Describe JJ Thomsons Cathode Ray Experiment. The discovery of cathode rays lead to the discovery of electrons : What are cathode rays Cathode rays are a stream of electrons following through vacuum tube. The three subatomic particles that constitute an atom are:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
